Study design
This is a single-center, randomized, prospective randomized controlled trial (RCT) study. This study has been approved by hospital ethics [The Second Affiliated Hospital of Zunyi Medical University, ID:YXLL (KY)-2022-017], conducted by the principles of the Helsinki World Medical Association Declaration, and registered with the Chinese Clinical Trial Registry (registration number: ChiCTR2300072409, date of first registration: 13/06/2023). After a basic assessment, all eligible participants were randomly assigned, drawn from envelopes by an assistant, to a study group, or a control group, with the assignment confidential to the study staff. Data were collected on the first day of the intervention and the day of discharge, see Fig. 1.
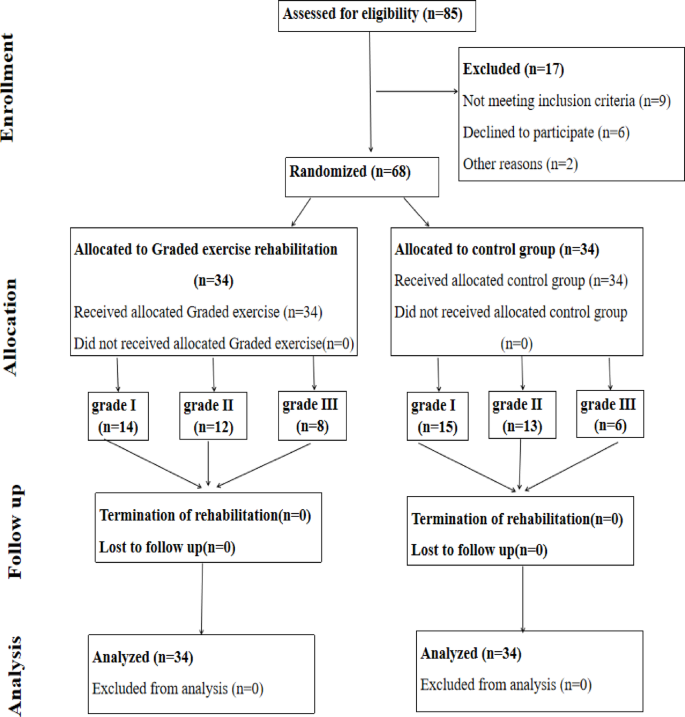
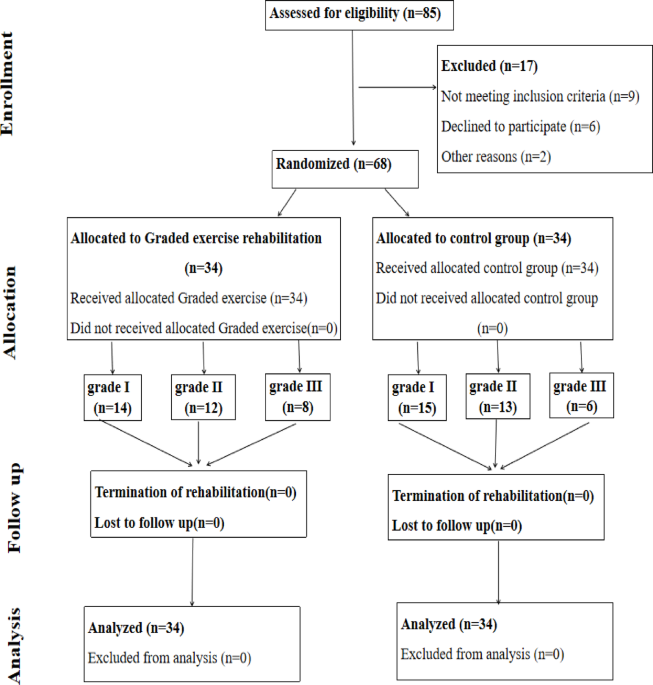
Consort diagram of the study.
Participants
68 AECOPD patients admitted to the Respiratory and Critical Care Department of a tertiary hospital in Zunyi City were included as the study subjects, they were assigned into 2 groups: the study group (n = 34) and the control group (n = 34). All participants provided written informed consent forms. The sample size estimation was based on the results of a previous study, and calculations were conducted using G*Power software. With an overall sample size of 54 participants, the total expected sample size was set at 68 participants to allow for a potential dropout rate of 20%.
The participants underwent screening based on the following inclusion criteria:(1) According to the diagnostic criteria for COPD in the Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2024 edition, and if the condition is in an acute exacerbation phase; (2) Meet the requirements of 2022 EXODUSEXpert consensus On Diaphragm Ultra Sonography in the critically ill, Expert consensus on severe diaphragmatic ultrasound detection: any one of the following: diaphragmatic mobility < 2 cm, diaphragmatic thickness < 2.2 mm, and diaphragmatic thickening fraction < 20%;(3) Age between 40 and 80 years old; (4) Clear consciousness, unobstructed language communication, and willingness to cooperate with the researchers in this study. The exclusion criteria were as follows: (1)There are contraindications for exercise: uncontrolled hypertension (> 180/110 mmHg) and hypotension (< 90/60 mmHg); Serious cardiovascular disease; Combined with acute attacks of bronchial asthma, pulmonary embolism, pneumothorax, etc.; There are diseases that affect motor nerves, muscles, bones, and joints; (2) Chest deformity, known diaphragmatic paralysis or chronic neuromuscular disease;
Randomization and blinding
The random allocation sequence was generated by computer in a 1:1 ratio, and an independent investigator graded patients who met the inclusion and exclusion criteria according to the severity of their disease and implemented allocation concealment through sealed opaque envelopes. A single-blind design was used, with random allocation sequence generation and intervention grouping performed by a researcher who was not involved in patient recruitment, and the sonographer and data analysts were unaware of the grouping information.
Intervention
Rehabilitation intervention starts on the second day of patient admission and ends on the day of discharge, with no time interval during the process, twice a day.
Establishing a study group
The research team consists of one nursing manager, one director of the respiratory department, one attending physician of the respiratory department, one B-ultrasound doctor, one rehabilitation doctor, one head nurse, and two respiratory nurses, as well as two nursing master’s students. The nursing manager serves as the team leader and is responsible for coordinating and allocating research work; The director of the respiratory department serves as the deputy team leader, responsible for AECOPD theoretical knowledge training and guidance; Respiratory doctors are responsible for diagnosing grading standards for AECOPD patients and ensuring patient safety during interventions; The B-ultrasound doctor is responsible for bedside diaphragm B-ultrasound testing; Rehabilitation doctors are responsible for providing guidance and training on graded exercise rehabilitation knowledge; The head nurse is responsible for team management, guidance on plans, and quality control work; The responsible nurse is responsible for implementing a graded exercise rehabilitation program under the guidance of a rehabilitation doctor. Nursing master’s students are responsible for collecting baseline data and evaluation indicators.
Method of classification
Due to the rapid and severe progression of AECOPD patients, most of them were unable to complete lung function tests. Therefore, in this study, 68 AECOPD patients who met the inclusion and exclusion criteria were divided into grades I, II, and III according to the grading criteria (see Table 1). Patients in the above three grades were randomly divided into a study group and a control group using a random method.
Intervention methods for the control group
Routine exercise rehabilitation based on symptomatic treatment: ① Turning over: Assist in turning over and changing to a comfortable position, every 2 h; ② Effective cough guidance: Guide the patient to take 5–6 deep breaths first, then keep their mouth open at the end of inhalation, cough continuously several times to bring phlegm to the throat, and then cough vigorously to expel the phlegm; ③ Clamping vibration: The palm is in an empty cup-shaped shape, tapping the lungs from bottom to top and from outside to inside. Each lung lobe is tapped for 1–3 min, lasting for 5–15 min, twice a day; ④ Mechanical sputum removal: Use a vibrating sputum removal device with a frequency of not less than 200 times/min, and the tremor sequence and duration are the same as those of manual lung tightening, 15–20 min, twice a day; ⑤ Pursed lip breathing: The inhalation time and exhalation time should be maintained at a ratio of 1:2 or more, with an expected breathing frequency of 8–10 breaths per minute. Each training session should last for about 5 min, twice a day; ⑥ Abdominal breathing: respiratory rate of 8–10 times/min, each training lasting about 5 min, 2 times/day; ⑦ Upper limb exercise: Dumbbell exercises, place 0.5 kg dumbbells on each hand at shoulder height, use the other hand to straighten and lift and lower the dumbbells, inhale when lifting, exhale when lowering, and repeat this exercise with the other hand. 3 sets/time, 8 pieces/time, 2 times/day; ⑧ Lower limb exercise: Sit in a chair, straighten your knees, keep your legs straight for 5 s, and then relax. Repeat this process, 3 groups/time, 8 groups/time, 2 times/day.
Intervention methods of the study group
Rehabilitation intervention starts on the second day of patient admission and ends on the day of discharge, with no time interval during the process, twice a day. Graded exercise rehabilitation based on symptomatic treatment. This exercise plan has been supported by evidence-based research and two rounds of expert consultation, ensuring its scientific and safe nature. The plan includes pre-exercise safety assessment, exercise intensity, exercise frequency and time, exercise adjustment principles, and suspension standards during exercise. The specific plan is shown in Table 2.
Observation and evaluation indicators
Evaluation time
Both groups of patients were tested and scored within 24 h after enrollment and on the day of discharge.
Evaluation indicators
-
1.
Diaphragm detection: in this study, a portable color Doppler ultrasound system (Mindray UMT-200, Shenzhen Mindray Bio-Medical Electronics Co., Ltd.,Shenzhen, China) was used to detect the right diaphragm area of patients. The low-frequency convex array probe was mainly used to observe diaphragm movement, and the high-frequency linear array probe was used to observe diaphragm thickness. All test values were obtained by the same ultrasound physician using the same measurement method. The detection method is detailed in Fig. 2.
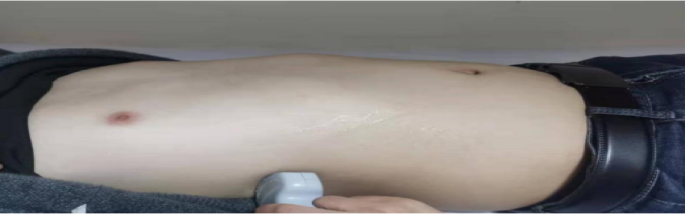
Bedside ultrasonic testing area (right side).
① Diaphragmatic mobility
Refers to the difference in displacement distance of the diaphragm at the end of exhalation and inhalation. The patient is in a supine position, with the convex array probe placed at the junction of the midline of the clavicle and the lower edge of the rib arch. The liver is used as the sound window, and the probe is perpendicular to the diaphragm dome. The ultrasound beam is directed toward the head and left and right scans are performed to locate the position of the diaphragm. On the B-mode ultrasound image, select the location with the highest motion amplitude, make the sampling line perpendicular to the diaphragm, convert it to M-ultrasound, and display the trajectory of the diaphragm along the sampling line with respiratory movement. Adjust the scanning speed according to the subject’s respiratory rate, record the difference in distance between the end-expiratory and end-inspiratory phases and the sampling line, measure three times, and take the average. See Fig. 3.
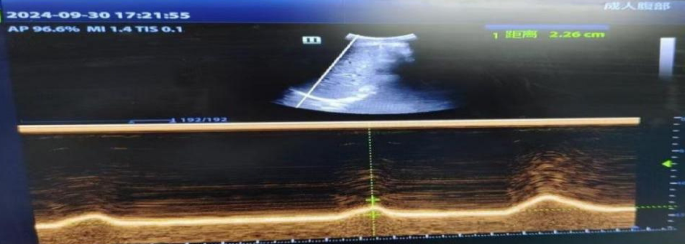
Schematic diagram of diaphragm movement measurement (right side).
② Diaphragm thickness and diaphragm thickening fraction
The patient is in a supine position, and a linear array probe with a frequency of 7–13 Hz is placed between the 7th or 8th rib of the right anterior axillary line, which is the area where the diaphragm meets the chest wall. The movement of the diaphragm is scanned, and the distance between the parallel high echo inner sides of the diaphragm on both sides is measured, which is the diaphragm thickness. Measure the end-inspiratory diaphragm thickness (DTei) and end-expiratory diaphragm thickness (DTee) under calm breathing for three respiratory cycles, and calculate the average value. Obtain the diaphragm thickening fraction DTF = (DTei DTee)/DTee × 100% through the formula. See Fig. 4.
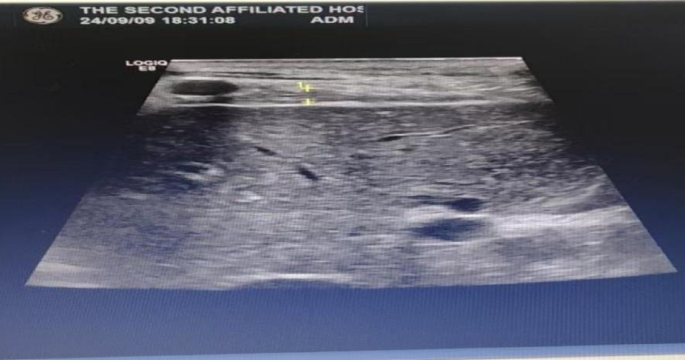
Schematic diagram of diaphragm thickness measurement (right side).
-
2.
Sports endurance: using a 6-min walk test (6 MWT) as the testing standard.
-
3.
Quality of life: assessed using the Chronic Obstructive Pulmonary Disease Assessment Test Score Scale (CAT).
-
4.
Respiratory condition: evaluated using the Modified Dyspnea Rating Scale (mMRC).
-
5.
Noninvasive mechanical ventilation duration and length of hospital stay.
-
6.
The overall incidence of complications related to non-invasive mechanical ventilation.
Statistical analysis
Statistical analysis was conducted using SPSS version 29.0 statistical software, and the measurement data followed a normal distribution expressed as mean ± standard deviation. A Paired t-test was used for intra-group comparison before and after, and two independent sample t-tests were used for inter-group comparison.
link